How Are Diamonds Made And Formed
Ever since diamonds were first discovered in South Africa, they have found their way into our culture as symbols of love, status and wealth.
While this can be credited to De Beers’s ingenuous marketing campaigns and famous slogan “A Diamond is Forever“, have you ever wondered where do diamonds come from?
Despite the significance they have in our culture, most people actually don’t know much about the precious stones that adorn the jewelry we wear.
In this writeup, we look at the different ways that diamonds are formed in nature and how diamonds are made using modern technology. We’ve also created a fully animated infographic to help you visualize what happens during the various processes.
Click Here to Jump to Animation
How Are Diamonds Formed Naturally on Earth?
Did you know that diamonds are made of carbon and it is fundamentally the same material as pencil lead (graphite)? Yet, they are the hardest substance ever known to mankind and have completely different properties as graphite.
What gives diamond their unique properties is the covalent bonding between its atoms.
You see, when diamonds are formed under extreme temperature and pressure, atoms in their crystalline structure become tetrahedrally bonded (each carbon atom links to 3 other carbon atoms) to create a strong and stable lattice.
Diamonds Are Formed in the Earth’s Mantle
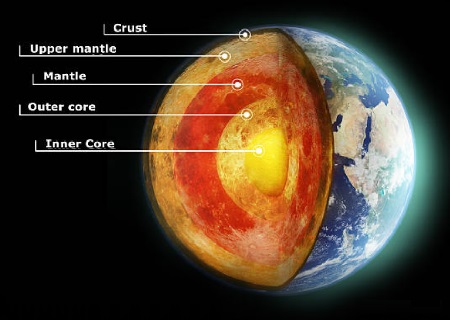
After years of research, geologists and scientists have a better understanding of how diamonds form in nature. It is believed that the right conditions for diamonds to be created are at elevated temperatures of 900°C to 1300°C in combination with an extremely high pressure of 650,000 psi to 850,000 psi.
In nature, there are only certain places that provide conditions like these and that’s deep within the Earth.
In fact, most diamonds form inside the Earth’s mantle under parts of the continental crust called cratons. These provide stable environments that allow diamond crystals to grow over millions of years before they make their way to the surface via Kimberlite pipes.
And it’s no coincidence that cratons are found in continents like Africa and Australia which also happen to be the biggest producers of gem-quality diamonds in the world.
Among the natural processes, this is the only method that produces diamonds large enough for jewelry as there is sufficient time for large, single-crystal stones of superior clarity and purity to develop.
Diamond Formation in Subduction Zones
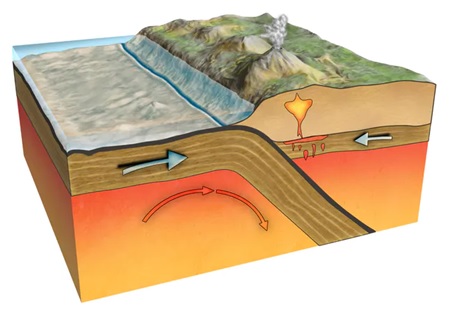
When tectonic plates collide, one massive land mass is forced underneath the other into an area known as the subduction zone. If one of these plates carry rocks and materials rich in carbon content, they melt under high temperature and pressure to create diamonds.
Interestingly, diamonds formed by this process are generally small in size (between 1–80μm) and not viable for commercial use. Some of them make it back to the surface due to plate movements while others may lie in place for millions of years before being brought to the surface via volcanic eruptions.
Diamonds Made by Asteroid Impacts

When an asteroid strikes the Earth’s surface, the epic collision creates a massive explosion of heat and pressure on the ground. Carbon based deposits in the impact crater can get turned into tiny diamonds in an instant.
The Popigai crater in Russia is a spot which was struck by a large asteroid millions of years ago. And the immense pressure and temperature generated by the impact turned the surrounding metamorphic rocks and graphite into diamonds (less than 2mm in size).
Diamonds Deposited by Meteorite Fall

Besides creating a spectacular sight in the night sky, meteorites also deposit their contents onto the ground when they hit the Earth’s surface. Researchers from NASA have detected large volumes of nanodiamonds in meteorites that originated from deep space.
In April 2018, the National Geographic even reported that some of these diamond-bearing meteorites could be leftover space rocks from remnants of early planets that never formed.
How Are Diamonds Made in a Lab By Man?
The first scientific breakthrough in man-made diamonds came in 1954 when General Electric developed a process that successfully replicated the conditions for natural diamond formation.
This process is called High Pressure High Temperature (HPHT) and involves the use of seed crystals which are grown in superheated anvils.
High Pressure High Temperature (HPHT)
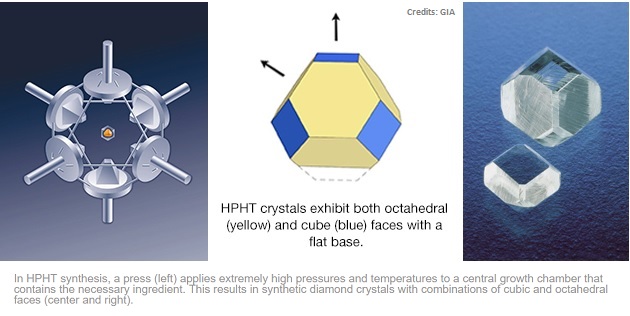
HPHT is a process that is energy intensive and has a relatively low production rate (seed crystal typically grows at a few microns per hour). During the process, the parameters need to be tightly controlled or it may cause the diamond’s growth to fail or the rough to become heavily included.
Chemical Vapor Deposition (CVD)
Chemical vapor deposition (CVD) is another method of making synthetic diamonds on a commercial scale. In this process, diamonds are grown in a hydrocarbon gas mixture in a controlled environment.
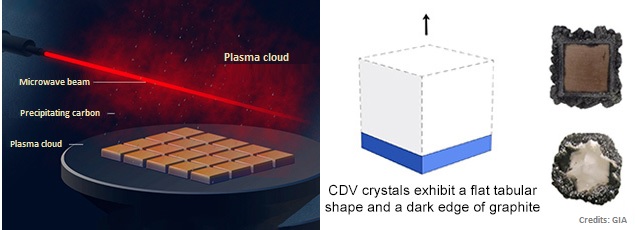
The hydrocarbon gas (usually methane, CH4) is heated to elevated temperatures and broken down into its atomic state. Next, the carbon atoms are then precipitated onto seed crystals and the rough diamond is grown layer by layer.
Compared to HPHT, the CVD method is relatively easy to setup and it is possible to grow synthetic diamonds over large surface areas at the same time. Besides lower temperatures, the CVD method doesn’t require high pressures and achieves higher growth rates than HPHT.
Conclusion
So there you have it, we’ve learnt how diamonds are made naturally and how they are created in the laboratory. But this is just the beginning of our understanding of this fascinating gemstone.
As scientists gain a better understanding of the physical world, I’m very sure they will discover more ways in which diamonds are made in nature. Also, as engineers push the boundaries of manufacturing research and development, I foresee a future in which synthetic diamonds can be made at lower costs and with better efficiency.
We truly live in exciting times. And I look forward to the years ahead where diamonds will be used as a super material to propel current technology to greater heights and drive high performance applications.
Animated Infographics by Beyond4Cs.com
To help you visualize the various formation processes, we’ve created this infographics for an easier understanding of the concepts. Check it out below and feel free to share or embed this infographics to your website using the code below…
Embed This Infographic On Your Site (copy code below):

Embed This Infographic On Your Site (copy code below):
Like the infographic and want to share it? Feel free to copy and paste the code in the box below to your website. If you need a higher resolution image file, feel free to get in touch and I’ll generate it for you.
References And Citations
Ulrika F.S. D’Haenens J.,Andrey K.,Kyaw S.M.,Paul J. & Wuyi W. (2015). Large Colorless HPHT Synthetic Diamonds from New Diamond Technology. Gems & Gemology, Fall 2015, Vol. 51, No. 3. Retrieved from https://www.gia.edu/gems-gemology/fall-2015-large-colorless-hpht-grown-synthetic-gem-diamond-technology-russia
Sumiya H., Toda N., Satoh S. (2005) Development of high-quality large-size synthetic diamond crystals. SEI Technical Review, Vol. 60, pp. 10–16.
Branko D., Matthias A., Adolf P., Mikko A. & Andrey K. (July 7, 2013). NDT Breaking The 10 Carat Barrier – World Record Synthetic Diamonds Investigated. Retrieved from http://gemresearch.ch/ndt-breaking-the-10-carat-barrier-world-record-faceted-and-synthetic-diamonds-investigated/.
Robert L. (April 30, 2013). CVD-Grown Synthetic Diamonds, Part 1: History. Retrieved from https://www.gia.edu/news-research-CVD-grown-part1
Shigley, J.E. (July 25, 2016). Identifying Lab-Grown Diamonds. Retrieved from https://www.gia.edu/identifying-lab-grown-diamonds
Yaakov W, John M, Graham D.P., Geoff M.N. & Chris J.O. (2015). Highly saline fluids from a subducting slab as the source for fluid-rich diamonds. Nature volume 524, pages 339–342 (20 August 2015). Retrieved from https://www.nature.com/articles/nature14857
Lawson S.C., Kanda H. (1993) An annealing study of nickel point defects in high-pressure synthetic diamond. Journal of Applied Physics, Vol. 73, No. 8, pp. 3967–3973. Retrieved from https://www.gia.edu/gems-gemology/spring-2004-identification-cvd-synthetic-diamond-martineau.
Other Interesting Reads
Ice-VII Inclusions in Diamonds: Evidence for Qqueous Fluid in Earth’s Deep Mantle – Science Mag
‘Diamond Rain’ Falls on Saturn And Jupiter – BBC
Is Diamond Really a Super Material? – Global Spec
Synthetic Diamonds: The Industrial Supermaterial – ECN
New Diamond Nanothreads Could be the Key Material for Building a Space Elevator – Extreme Tech
Using Diamonds to Create Drug Discovery Tools – Technology Networks
Diamonds Are Made From Recycled Seabed – Cosmos Magazine
Leave A Comment







16 Comments
I am very curious about what are diamonds made of when the ones I had seen could glow in the dark. I have found several rough specimens in a nearby quarry and would like to sell them. Some are as large as 10 carat in size. Can you help?
Diamonds are made of carbon with some trace elements as impurities. Trace elements like boron, nitrogen or aluminum can change the behavior of the diamond to light. The phenomenon you are describing is phosphorescence which causes the material to emit light after a source is removed. And just to be sure, it rarely occurs in real diamonds. Now, not to sound condescending, if what you really found are diamonds and they are so easily available in your nearby quarry, I doubt you would be asking me basic questions here. In fact, I doubt the roughs you have in hand are really diamonds.
When did diamonds form and how long does it take in nature to do that?
With our current understanding, it’s hard to pinpoint an exact date or how long it takes to make a diamond inside the mantle. Based on studies and scientific data, it is suggested that the oldest diamonds are between 1 billion to 3 billion years old and the time taken to make them could be anywhere between these 2 figures since the process is intermittent.
For example, a diamond could form partially over the span of millions of years only to lay dormant because conditions aren’t right. When conditions turn favorable again, the growth of the diamond crystal could continue. And that’s one part of the diamond formation process that scientists don’t fully understand yet.
With carbon dating technology being so advanced, why is it that scientists cannot date the age of diamonds accurately?
Carbon dating can work on the inclusions found in the diamonds but not on the diamonds itself. So, any dating data collected only gives us a rough idea and not an accurate result of the actual age of the diamond formation. This link has some further explanations: http://grisda.org/resources1/faq/radiocarbon-dating/
It’s the same issue with trying to date the history of the surrounding minerals and materials. It only gives a gauge of the timeline in diamond creation but not a precise accounting.
Do you have more details on how long does it take for a diamond to form via CVD from the start till the end?
It depends on different process recipes and what is the kind of synthetic diamond you are targeting to produce. To be honest, some of the recipes are trade secrets and I don’t have all the exact details. What I would say is that the duration of growth can take place from a few days to a 4 weeks.
This handbook from Element 6 provides a lot of details for reference: https://e6cvd.com/media/wysiwyg/pdf/E6_CVD_Diamond_Handbook_A5_v10X.pdf
Very interesting animation and really informative! I’ve read quite a number of topics on the subject and came across one that said the outer planets could be full of diamonds. It’s still a mystery as to why they are there and the universe is full of surprises.
Some astronomers believe that it actually rain diamonds in the atmospheres of gas planets like Jupiter and Saturn. If you are wondering how are diamonds formed in space or in other parts of the solar system, this article from Business Insider puts forward some theories. And yes! It’s really fascinating to think about what lies beyond our planet. I’m a huge fan of “The Universe” documentary series from The History Channel and I would recommend that to people who have similar interests as well.
I’ve been looking around for resources on how diamonds are formed for kids. Can I get your permission to republish this article in our newsletter?
I’m glad you found it useful. You are welcome to repost it as long as attribution and credits are given back to Beyond4cs.com. There are several other pages on Beyond 4cs on topics that touch upon how are synthetic diamonds made and identified. They might be worth checking out as well.
Thanks Paul
Your work looks very interesting.
I’ll have to call you the King of Diamonds!
Regards
Ric Johnson
I have always found your articles on diverse mining and minerals interesting and informative.
I wonder if you assist producers and traders in rough or polished gemstones from Africa to find appropriate buyers in Asia, Europe, Australia and USA?
Dear Paul,
Thank you so much for your most informative newsletters. Your research, and love of diamonds leaves me speechless.
Sincerely, Anne
Unfortunately, I don’t offer such a service and I don’t have the proper knowledge or contacts to legally do this.