The Formation Process of Diamonds in Nature

The covalent bonding of carbon atoms makes diamond the hardest substance.
For most people, the journey from the day of your marriage proposal to your actual wedding day kiss was probably longer than what you would have imagined.
Now, if we were to go backwards step-by-step from the wedding kiss, we would probably see the fretting and struggle of a guy trying to choose a perfect engagement ring.
And if we further interpolate the steps required to get a polished diamond from a rough, you could imagine the complex financial process that goes on between different members of the trade, the sweaty hands of miners working to bring the roughs to the surface, and so on and so forth.
For the majority of us, learning about the process in which diamonds come into existence is a much more interesting topic to enhance your general knowledge. In this article, let us take a look at how diamonds are formed in nature.
“Carbon” And “Coal” Are Related, But They Are Different

Low grade yellowish rough diamonds.
Carbon is, without any doubt, the most important chemical element in the universe. It takes on many forms and appearances, out of which diamond is the one that is most valuable. The others include a myriad of organic and inorganic substances.
Pure carbon on its own, has three major forms of appearance – coal, graphite and diamond.
From a chemist’s point of view, these three materials are identical as they contain the same type of atoms. The differences lie within their molecular structures which gives them their unique material and physical properties.
Now, you might have heard of an urban legend which associates diamond formation with coal formation. This myth states that diamonds are actually pieces of coal which had been transformed under high pressure and temperature.
Yet, science has proved this wrong. We now know this cannot be true because coal is formed from early vegetation and their remains. On the other hand, most of the diamonds we use today are much older than these vegetations that make up coal.
So, How Are Natural Diamonds Formed And Created?
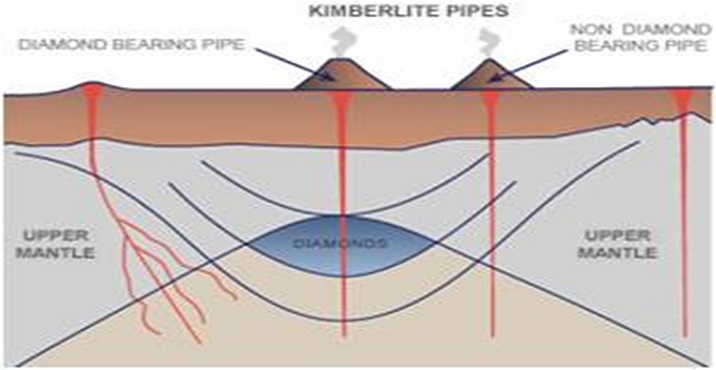
The majority of diamond deposits found in commercial mines were formed deep within the Earth’s mantle. The conditions for diamonds to form are very specific (you can think of it as a baking recipe) and it takes place in an area called “diamond stability zone”.
Diamond stability zones are usually located in the upper mantle of the Earth estimated to be around 150km below the Earth’s crust. It is an environment with high temperatures (>1000 degrees Celsius) and pressure (>725,000 pounds per square inch).
After formation, the diamond deposit could stay in the same location for millions of years until something catastrophic takes place. Through violent volcanic eruptions, deposits can be brought to the surface in large chunks of diamond-containing rocks called xenoliths. These eruptions also create pipes in the upper layers of Earth’s crust called kimberlite pipes.
Interestingly, not all kimberlite pipes contain diamond-bearing ore and a huge amount of random factors have to come together for that to occur. Even if a kimberlite pipe is found to contain diamonds, prospectors have to survey the ore intensively for economic viability before any large scale extraction could even begin.
This short video from Nat Geo sums up the formation process in a visual manner. Check it out!
Other Places Where Diamonds Are Created By Forces of Nature
There are a number of scenarios where diamond formation is possible. The most interesting out of these is probably the formation of diamonds in the outer reaches of our solar system. NASA researchers have found very tiny pieces of diamonds in extraterrestrial bodies, which have formed either in space or in the mantle of some other planets.
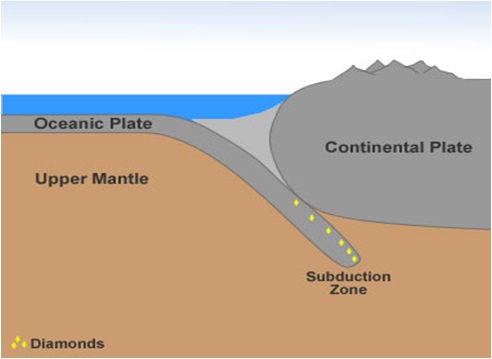
Also, there do exist obscure places on our planet which are ideal for diamond formation. For example, diamonds were discovered in subduction zones where two different tectonic plates meet each other.
Interesting Videos About Diamonds Worth Watching
Recently, I came across a couple of really interesting clips about where it rains diamonds, literally. You also get to go behind the scenes to see what happens in a diamond sorting center.
Diamonds are a byproduct of some very interesting processes in nature. Besides occurring in many types of shapes, colors and sizes, there are some cool looking features on a rough diamond you probably never seen before.
On the next page, I’ll show you interesting birthmarks found on rough diamonds called trigons. These are features that are found on a rough diamond’s skin when they are extracted from the ground.
Related Articles
Leave A Comment










2 Comments
Good. Very interesting video clip. I wonder how is a diamond formed on a planet like Saturn and where they would have different properties compared to those on Earth.
I would love to know as well. On Earth, when there are impurities or nitrogen centers in the diamond, it creates a phenomenon known as fluorescence. I wonder if those created in outer space have different features to them or not.